The adventures of Max the microorganism
INFOSHEET
The Adventures of Max the Microorganism is a visual story to support a positive and strong narrative about the application and beneficial uses of microorganisms and microbial products in our daily lives.
Max is a fictional character that embodies a microorganism embarking on a journey to help humans cope with the most pressing global challenges.
Like all microorganisms, Max is invisible to the human eye and was one of the first living creatures on Earth.
Max belongs to a large family of single or multi-celled organisms that includes bacteria, fungi, and algae. Indeed, microorganisms live in oceans, forests, plants, animals, and in human bodies.
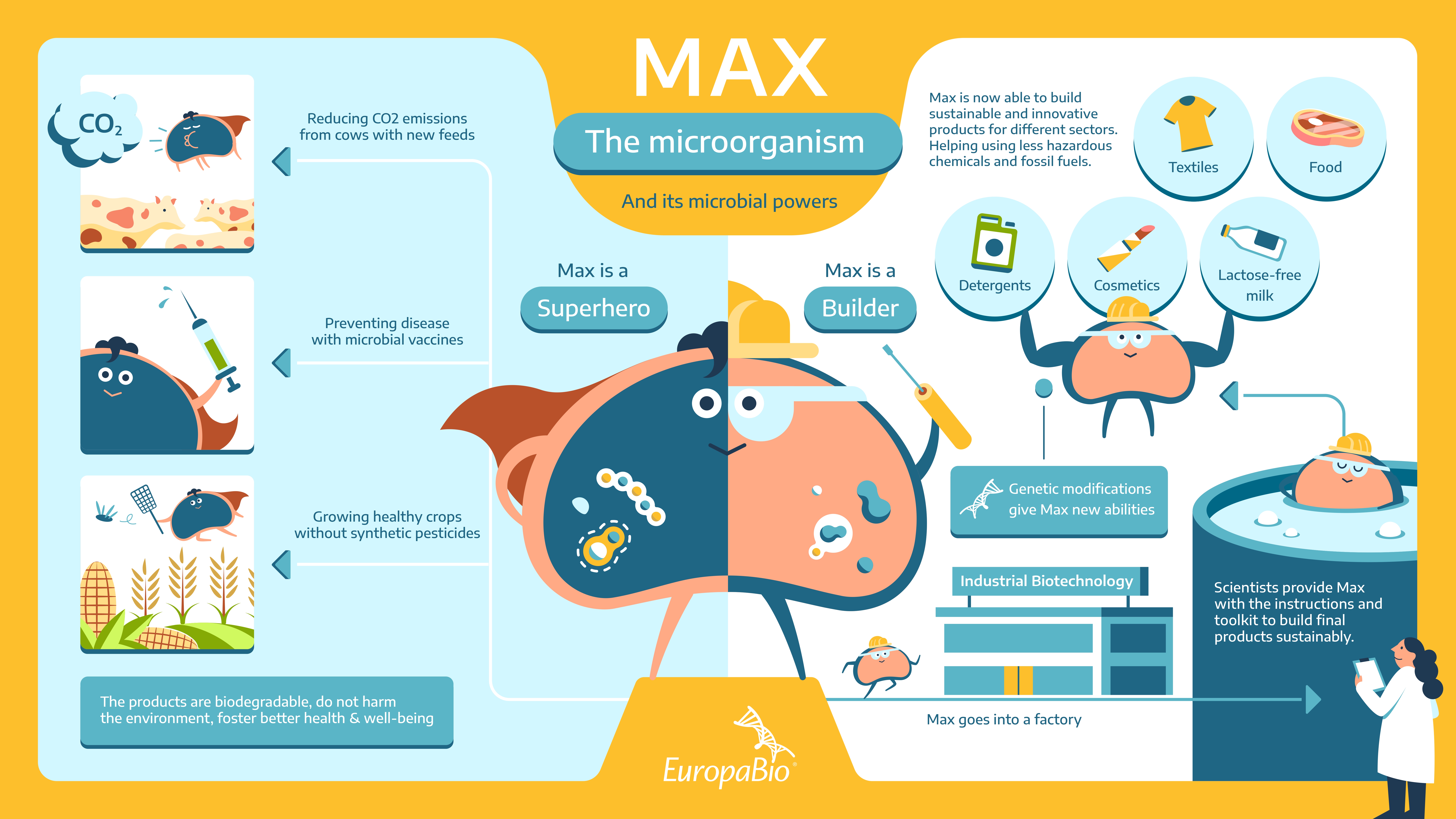
The journey of Max articulates as a builder and a superhero to showcase the different uses and applications of its microbial powers.
Industrial biotechnology and scientists provide Max with the right information and tools to produce many kinds of products. After a series of genetic modifications, Max becomes a builder able to produce its microorganisms-derived products. These include food (bread, beer, cheese), pharmaceuticals (insulin), consumer products (detergents, cosmetics), biofertilizers, and biopesticides. Indeed, microorganisms and their derived products act like building blocks for other industries, including biofertilizers and biopesticides.
As a builder, Max acquires all the necessary expertise in the field of biotechnology that allows it to develop alternatives to fossil-fuel-based products that don’t rely on hazardous chemicals. Max has unlocked its full microbial superpowers! As a superhero, Max helps humans optimize their quality of life in different sectors through microbial products, which are sustainable alternatives and beneficial to both the environment and humans’ health & well-being.
For example, Max, helps grow crops, and fights pests without harming biodiversity and destroying crop yields.
Max’s superpower is not limited to the agri-food sector. It can also use its resources to develop microbial vaccines, like the one against cholera.
The short adventures of Max highlight how microbial products and innovative biotechnology solutions are a win-win both for the environment and humans.



