European Week Against Cancer 2022 – Interview with Santiago Aguirre, Market Lead Europe Oncology, Sanofi

INTERVIEW


1) How is Sanofi using innovative healthcare biotechnologies to improve treatments for cancer patients?
We are building on a rich legacy in oncology. Our strong pipeline today reflects our renewed commitment to developing new therapies that could both change outcomes and have a positive impact on the lives and wellbeing of people living with many different types of cancer.
We are building a new generation of medicines designed to either attack and destroy cancer cells directly or mobilize the immune system to mount a strong defense against cancer. We have been reinforcing our foundations in disease biology with a diverse toolbox of advanced technologies for designing new types of medicines, and with strategic collaborations that help us advance the science more quickly. Because the more “weapons” for attacking cancer we stock in our armory, the more opportunities our scientists have for discovering therapies that will help patients.
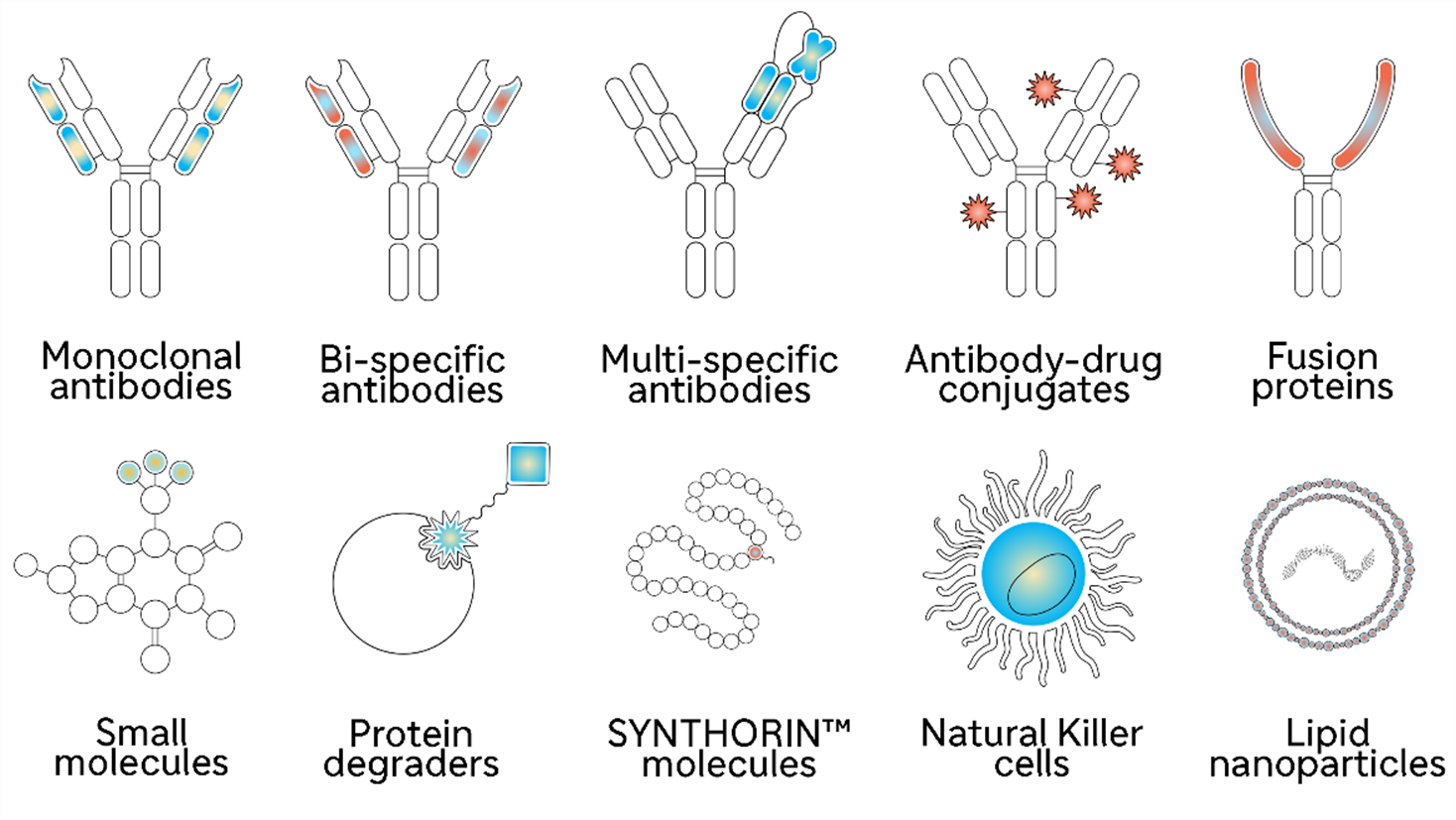
Breakthrough science has accelerated the development of innovative medicines, but patient access can lag when there are local barriers. The way local healthcare systems evaluate new cancer therapies and determine their value can contribute to delayed or restricted patient access.
Those who make budget decisions about patient access (‘payers’) have a strong preference for overall survival (OS) data as a measure of a cancer medicine’s value. Regulatory and reimbursement evaluations give limited weight to health-related quality of life (HRQoL) and patient reported outcomes. A lack of immediate OS evidence can delay a patient’s access to a new medicine that could offer added clinical benefit. It could even limit or even deny access for certain patient sub-populations.
“Patient relevant endpoints” include a wide and growing range of direct and indirect measures of clinical effectiveness beyond OS and HRQoL, such as progression-free survival and minimal residual disease. Stakeholders must work together to define frameworks and methods to support the application and acceptance of novel endpoints, as they present an opportunity to accelerate the research and development of new therapies and, consequently, the time to patient access.
3) What opportunities could the upcoming review of the EU’s pharmaceutical legislation bring to support the fight against cancer and improve access to new treatments?
Our purpose is to improve people’s lives. We work with health authorities directly and through our partners to ensure our products get to the patients who need them across the European Union (EU).
The revision of the EU marketing authorization framework announced by the European Commission represents an opportunity to improve EMA product filing conditions, and access to medicines. Today, only 1 in 5 new treatments originate in the EU; almost half of them come from the US. Since the 1990s, R&D investment in the EU has grown 5 times compared to 10 times in the US: a reversal of the situation 25 years ago.
We support the European Commission’s ambition to modernize the EU marketing authorization process, adapt it to advances in science and technology, and address societal expectations to enable greater and faster access to innovative therapies. This is particularly important for therapies addressing life threatening cancers.



