EuropaBio NAC Summit 2022: key recommendations on how National associations can deliver Europe’s innovation, investment and excellence in biotechnology

PRESS RELEASE
National Associations Council (NAC) Summit is a yearly EuropaBio members occasion to discuss the emerging issues which can have an impact on the national and EU level to the sector. This year, the Summit addressed Europe’s Industrial Strategy and how biotechnology sector can contribute to its delivery across the sectors.
The discussions at the Summit were summarised in the key policy recommendations at national and the EU level which could serve the policymakers to get a better understanding of the sector’s potential and regulatory challenges in operating on the national and EU level.
Recommendations for Healthcare biotechnology policies
Developing novel medicines from biotechnology like mRNA and cell & gene therapies is a long-term, high-risk venture which needs an agile business environment in Europe. We need a regulatory system that is accessible for SMEs, given a lot of innovative medicines originate from SME biotechnology companies. When compared globally Europe is not sufficiently competitive in terms of overall timelines for review and approval of regulatory submissions. The world is moving and Europe needs to build a critical mass in target technologies and industries if it is to be a producer rather than a customer in the coming decades.
‘The European system of medicines regulation needs to become more streamlined, with expedited pathways to accelerate approval of innovative new treatment. An agile regulatory environment will bring the EU in line with other regions in the world, bringing patients access to innovative medicines earlier.’ said Sean Byrne, Europabio.
When it comes to clinical trials, one big struggle is the lack of interoperability of health data. Increased digitalisation could support effort to reduce duplication, and reduce administrative burden. There should be equal opportunity for industry, regulators, HTA agencies, payers and other stakeholders to reuse important information and facilitate relevant applications.
‘If you have different protocols, different ways of using data, different interpretations of GDPR, it's very hard to use this data in a cross-border setting, which covers different member states. We are waiting for the Health Data Space Regulation, the proposal which should resolve this issue. And of course we believe that when this is done, this will be a big step forward.’ said MEP Tomislav Sokol.
There’s huge potential in getting cross-border healthcare right; it delivers for patients and can assist in addressing issues around access to medicine. The European Reference Networks are particularly important for two reasons: 1) better access for rare disease patients, and leveraging the network of excellent physicians across Europe, 2) the potential to work with industry to develop clinical trials and further R&D.
‘A European reference networks that are established and are working really well, probably should be upgraded in terms of their responsibilities and activities and involvement also in cross-border health. And also the physicians community needs to have a mindset change to make sure that they treat their patients potentially in a different setting, i.e. across borders.’, said Thomas Bols, PTC Therapeutics
.Full list of recommendations:
• Create regulatory frameworks suited to accelerating novel technologies to market
• Align the information required for regulatory and HTA frameworks
• Review instruments such as state aid to enhance European competitiveness globally for higher-risk investment
• Enhance national cooperation for alignment of data to enable greater international benefit
• Improve timelines and procedural transparency for patients to access to advanced novel therapies and healthcare across Europe
• Promote industry engagement to strengthen the ability of ERNs within research and innovation for patient benefit

The EU Green Deal has set an important sustainability framework and path towards climate neutrality for the EU. This comes with opportunities as well as challenges, and both new and existing solutions will be needed.
‘It is important to have a European Green Deal that supports biotechnology research and innovation but also to concretely encourage the use of bio-based alternatives in relevant sectors such as packaging, chemicals, coatings, fertilisers, cosmetics etc. At the same time, it is important to ensure that new innovative products can come to market in a timely manner. The potential of market stimulation measures for bio-based products has still not been realised.’, said Agnes Borg, EuropaBio.
Europe needs for a science-based, proportionate and predictable regulatory approach to current and future biotechnology innovation. ‘Investing into research and innovation and setting ambitious policy objectives are very important, but not enough. Innovative products and breakthrough innovations could bounce back on the reality of fence. European regulatory frameworks and objectives need agile regulatory and policy frameworks to speed up time to market.’, said Elke Duwenig, BASF.
One concrete example is the current discussions around the EU GM regulatory framework, which is not fit for purpose for new genomic techniques and their products.
‘We have a problem in Europe with our regulatory framework, and the GMO legislation is really hampering innovation. And although there is the modernisation in progress now, if they only include plant biotechnology and only plant entities, then we won't make it. We must include microorganisms as well in other biotech applications.’, said Wieteke Wouters, HollandBio.
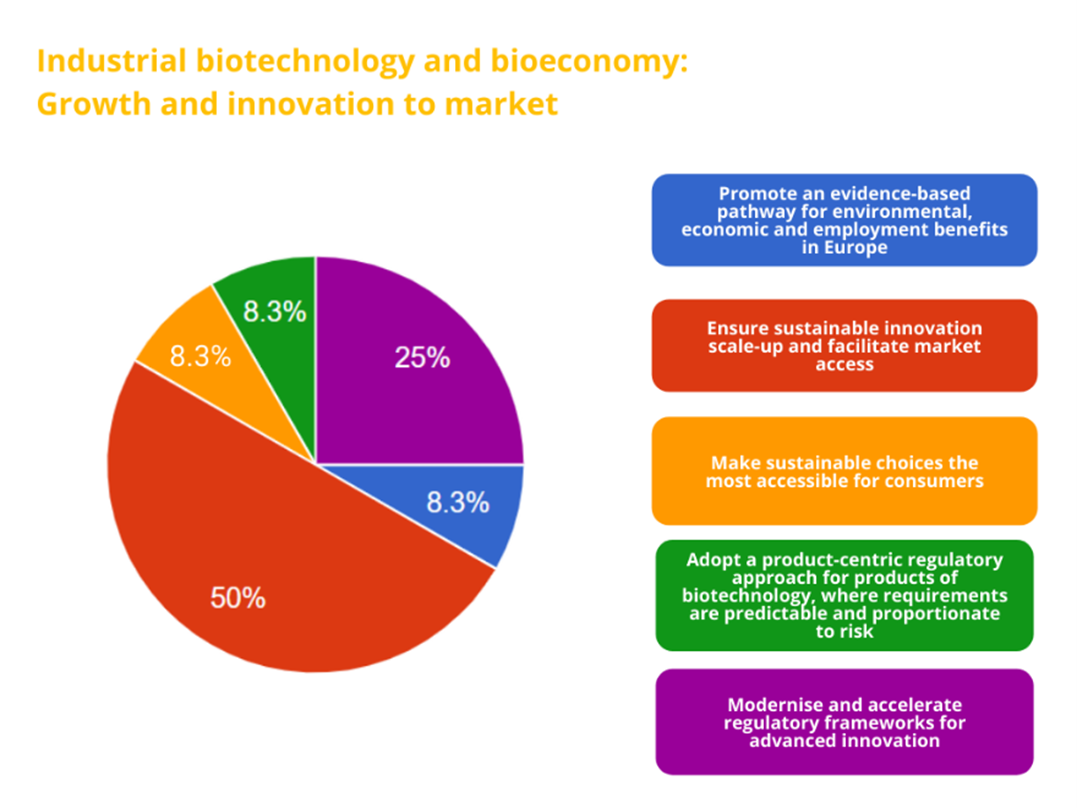
Full list of recommendations:
• Promote an evidence-based pathway for environmental, economic and employment benefits in Europe
• Ensure sustainable innovation scale-up and facilitate market access
• Make sustainable choices the most accessible for consumers
• Adopt a product-centric regulatory approach for products of biotechnology, where requirements are predictable and proportionate to risk
• Modernise and accelerate regulatory frameworks for advanced innovation